Coordination environments and π-conjugation in dense lithium coordination polymers†
Abstract
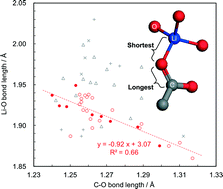
The understanding of lithium–oxygen coordination systems is important for making better lithium conductors as well as active materials for lithium ion batteries. Here, we report a systematic investigation on coordination environments in lithium coordination polymers (LCPs) through the syntheses and analyses of six new crystals composed of lithium ions and anthraquinone (aq) derivative anions, where the negative charges are distributed in π-conjugation systems. Their structures were determined by single-crystal X-ray diffraction to be (1) [Li2(23dcaq)(H2O)] in space group P21/c, (2) [Li(23dcaqH)] in P21/c, (3) [Li2(15dhaq)(H2O)2] in P21/c, (4) [Li2(14dhaq)(H2O)2] in Pnma, (5) [Li(14dhaqH)(H2O)] in P212121 and (6) [Li(14hnaq)(H2O)] in P212121 (23dcaq2− = 2,3-dicarboxy-aq, 14dhaq2− = 1,4-dihydroxy-aq, 15dhaq2− = 1,5-dihydroxy-aq and 14hnaq− = 1-hydroxy-4-nitro-aq). Through the comprehensive structure analysis of these materials as well as other LCPs, we found that when considering the longest C–O bond in the π-conjugation system of an anionic organic molecule and its coordination to a Li ion, there is a weak inverse relationship between the C–O and Li–O bond lengths. In addition, despite exhibiting optical band edges below 2 eV and 1D π-stacking connectivity, conductivity measurements on single crystals of 1–6 confirmed that they are all electronic insulators. We rationalize this finding on the basis of π-orbital delocalization, which is more restricted in the aq-based LCPs compared to known semiconducting hybrid materials.
- This article is part of the themed collection: In celebration of Tony Cheetham’s 70th birthday

 Please wait while we load your content...
Please wait while we load your content...