A fast click–slow release strategy towards the HPLC-free synthesis of RNA†
Abstract
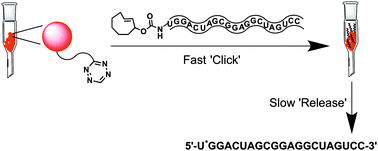
A general strategy for purification of oligonucleotides synthesized by solid phase synthesis is described. It is based on a recently developed concept involving a bio-orthogonal inverse electron demand Diels–Alder reaction between trans-cyclooctene and tetrazine, termed ‘click-to-release’. The strategy has been applied towards the synthesis and purification of a model hairpin RNA strand, as well as a 34 nt long aptamer.

 Please wait while we load your content...
Please wait while we load your content...