Comparison of ion mobility-mass spectrometry and pulsed-field gradient nuclear magnetic resonance spectroscopy for the differentiation of chondroitin sulfate isomers†
Abstract
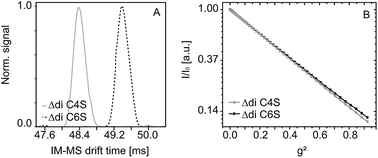
Sulfated glycosaminoglycans such as chondroitin sulfate (CS) are important, natural polysaccharides which occur in many biological tissues and possess different biotechnological and medical applications. The analysis of CS is normally based on the evaluation of the unsaturated disaccharides, which can be easily obtained by enzymatic digestion. The 4-sulfate and the 6-sulfate are the typical main products which are usually differentiated and quantified by MS/MS. Here we show that both disaccharide isomers can be readily differentiated by ion mobility-mass spectrometry (IM-MS), with the 6-sulfate isomer having a lower mobility in the gas phase. These findings are further substantiated by pulsed-field gradient (PFG) NMR, which also indicates lower mobility of the 6-sulfate isomer in solution. Based on these already well-studied isomers we provide evidence that the combination of both methods, IM-MS and (PFG) NMR, allows the differentiation of isomers in mixtures, the determination of the relative contributions of both isomers and the structural identification without the absolute need for pure, internal standards.

 Please wait while we load your content...
Please wait while we load your content...