Electrochemical sensing of nitro-aromatic explosive compounds using silver nanoparticles modified electrochips
Abstract
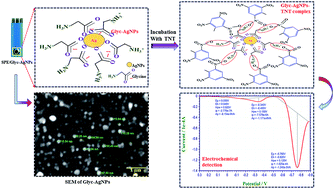
In the present study an electrochemical deposition method for glycine-stabilized silver nanoparticles (Glyc-AgNPs) at screen printed electrodes is shown in a single step with minimal sample preparation. The advantage of this method is the direct assembly of Glyc-AgNPs at the electrode surface without external modification. The Glyc-AgNPs were characterized for their size, crystallinity and redox activity. The nanoparticles were further used for electrochemical sensing of 2,4,6-tri-nitro toluene (TNT) and di-nitro benzene (DNB), i.e. nitro-aromatic explosive compounds using differential pulse voltammetry (DPV). The Glyc-AgNP modified electrochip revealed significantly more sensitivity towards TNT as compared to DNB, as evident from the current intensity obtained due to the electrochemical reduction process. Earlier studies have suggested that tri-nitro compounds are more easily reduced as compared to mono- and di-nitro compounds, which is hypothesized as the main reason for the observed sensitivity for TNT. A linear response was observed in the range from 1 × 10−10 to 0.1 M for TNT, and 1 × 10−7 to 0.1 M for DNB. The results obtained with spiked samples provided solid grounds for future field trials with the developed sensing device.

 Please wait while we load your content...
Please wait while we load your content...