Identification of human chorionic gonadotropin hormone in illegally distributed products by MALDI-TOF mass spectrometry and double-injection capillary zone electrophoresis
Abstract
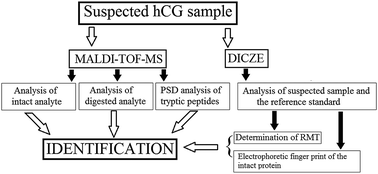
This paper describes two complementary methods, based on peptide-mass fingerprinting (PMF) using MALDI-TOF mass spectrometry and double injection capillary zone electrophoresis for identification of human chorionic gonadotropin (hCG). The identification was performed by determining the amino acid composition and comparing measured mass spectra with those predicted in silico. Capillary zone electrophoresis in double injection mode (DICZE) was used to confirm the identity of the protein. In DICZE, the identification was performed by analyzing illegal samples together with the corresponding reference standard during a double injection capillary zone electrophoretic run. The identification was based on the closeness of agreement between the calculated migration time (tmig(c)) and the observed migration time (tmig) of the reference standard. Furthermore, the DICZE method provides the possibility to compare the electrophoretic separation patterns of the native reference standard and the target analyte. The inner surface of the capillary was dynamically coated with putrescine, present in the BGE at 8.2 mM, to improve the separation. The CZE analyses revealed that the protein consisted of at least ten glycosylated isoforms.

 Please wait while we load your content...
Please wait while we load your content...