A multi-responsive turn-on flurogenic probe to sense Zn2+, Cd2+ and Pb2+: left-right-center emission signal swing†
Abstract
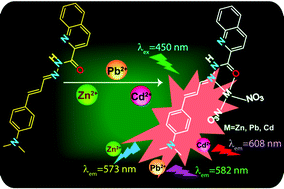
A versatile new fluorogenic Schiff base probe (L) has been synthesized by the reaction of quinoline-2-carbohydrazide (which acts as the chelating site) and 4-dimethylamino cinnamaldehyde (which acts as the signaling unit). L can sense three of the most biologically and environmentally important metal ions, Zn2+, Cd2+ and Pb2+, among various tested metal ions through selective TURN-ON fluorescence responses in physiological pH. Interestingly, L can not only sense Zn2+, Cd2+ and Pb2+ fluorometrically in physiological conditions but can also distinguish one from another by exhibiting individual intrinsic left-right-center TURN-ON emission signal swings. These selective fluorescence responses were explained by a chelation-enhanced fluorescence (CHEF) mechanism. Theoretical calculations were carried out to ascertain the preferred L–metal ion binding mode.

 Please wait while we load your content...
Please wait while we load your content...