Quantitative analysis of trace palladium contamination in solution using electrochemical X-ray fluorescence (EC-XRF)†
Abstract
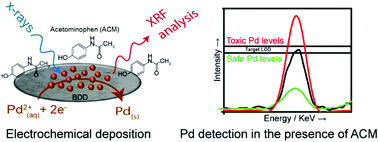
The application of electrochemical X-ray fluorescence (EC-XRF), for the detection of palladium (Pd) contamination in a range of solutions containing electrochemically active compounds, present in excess and relevant to the pharmaceutical and food industries, is reported. In EC-XRF, EC is used to electrochemically pre-concentrate metal on an electrode under forced convection conditions, whilst XRF is employed to spectroscopically quantify the amount of metal deposited, which quantitatively correlates with the original metal concentration in solution. Boron doped diamond is used as the electrode due to its very wide cathodic window and the fact that B and C are non-interfering elements for XRF analysis. The effect of several parameters on the Pd XRF signal intensity are explored including: deposition potential (Edep), deposition time (tdep) and Pd2+ concentration, [Pd2+]. Under high deposition potentials (Edep = −1.5 V), the Pd XRF peak intensity varies linearly with both tdep and [Pd2+]. Quantification of [Pd2+] is demonstrated in the presence of excess acetaminophen (ACM), L-ascorbic acid, caffeine and riboflavin. We show the same Pd XRF signal intensity (for [Pd2+] = 1.1 μM and tdep = 325 s) is observed, i.e. same amount of Pd is deposited on the electrode surface, irrespective of whether these redox active molecules are present or absent. For tdep = 900 s we report a limit of detection for [Pd2+] of 3.6 ppb (34 nM). Even lower LODs are possible by increasing tdep or by optimising the X-ray source specifically for Pd. The work presented for Pd detection in the presence of ACM, achieves the required detection sensitivity stipulated by international pharmacopeia guidelines.
- This article is part of the themed collection: Celebrating the 2016 RSC Prize and Award Winners

 Please wait while we load your content...
Please wait while we load your content...