Quantification of tunicamycin-induced protein expression and N-glycosylation changes in yeast†
Abstract
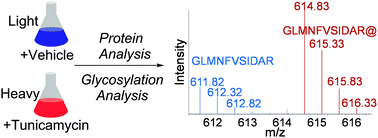
Tunicamycin is a potent protein N-glycosylation inhibitor that has frequently been used to manipulate protein glycosylation in cells. However, protein expression and glycosylation changes as a result of tunicamycin treatment are still unclear. Using yeast as a model system, we systematically investigated the cellular response to tunicamycin at the proteome and N-glycoproteome levels. By utilizing modern mass spectrometry-based proteomics, we quantified 4259 proteins, which nearly covers the entire yeast proteome. After the three-hour tunicamycin treatment, more than 5% of proteins were down-regulated by at least 2 fold, among which proteins related to several glycan metabolism and glycolysis-related pathways were highly enriched. Furthermore, several proteins in the canonical unfolded protein response pathway were up-regulated because the inhibition of protein N-glycosylation impacts protein folding and trafficking. We also comprehensively quantified protein glycosylation changes in tunicamycin-treated cells, and more than one third of quantified unique glycopeptides (168 of 465 peptides) were down-regulated. Proteins containing down-regulated glycopeptides were related to glycosylation, glycoprotein metabolic processes, carbohydrate processes, and cell wall organization according to gene ontology clustering. The current results provide the first global view of the cellular response to tunicamycin at the proteome and glycoproteome levels.
- This article is part of the themed collection: Emerging Investigators

 Please wait while we load your content...
Please wait while we load your content...