On the structural denaturation of biological analytes in trapped ion mobility spectrometry – mass spectrometry†
Abstract
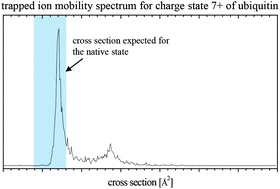
Key to native ion mobility/mass spectrometry is to prevent the structural denaturation of biological molecules in the gas phase. Here, we systematically assess structural changes induced in the protein ubiquitin during a trapped ion mobility spectrometry (TIMS) experiment. Our analysis shows that the extent of structural denaturation induced in ubiquitin ions is largely proportional to the amount of translational kinetic energy an ion gains from the applied electric field between two collisions with buffer gas particles. We then minimize the efficiency of the structural denaturation of ubiquitin ions in the gas phase during a TIMS experiment. The resulting “soft” TIMS spectra of ubiquitin are found largely identical to those observed on “soft” elevated-pressure ion mobility drift tubes and the corresponding calibrated cross sections are consistent with structures reported from NMR experiments for the native and A-state of ubiquitin. Thus, our analysis reveals that TIMS is useful for native ion mobility/mass spectrometry analysis.
- This article is part of the themed collection: Emerging Investigators

 Please wait while we load your content...
Please wait while we load your content...