Making the invisible visible: improved electrospray ion formation of metalloporphyrins/-phthalocyanines by attachment of the formate anion (HCOO−)†
Abstract
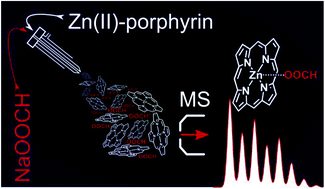
A protocol is developed for the coordination of the formate anion (HCOO−) to neutral metalloporphyrins (Pors) and -phthalocyanines (Pcs) containing divalent metals as a means to improve their ion formation in electrospray ionization (ESI). This method is particularly useful when the oxidation of the neutral metallomacrocycle fails. While focusing on Zn(II)Pors and Zn(II)Pcs, we show that formate is also readily attached to Mn(II), Mg(II) and Co(II)Pcs. However, for the Co(II)Pc secondary reactions can be observed. Upon collision-induced dissociation (CID), Zn(II)Por/Pc·formate supramolecular complexes can undergo the loss of CO2 in combination with transfer of a hydride anion (H−) to the zinc metal center. Further dissociation leads to electron transfer and hydrogen atom loss, generating a route to the radical anion of the Zn(II)Por/Pc without the need for electrochemical reduction, although the Zn(II)Por/Pc may have a too low electron affinity to allow electron transfer directly from the formate anion. In addition to single Por molecules, multi Por arrays were successfully analyzed by this method. In this case, multiple addition of formate occurs, giving rise to multiply charged species. In these multi Por arrays, complexation of the formate anion occurs by two surrounding Por units (sandwich). Therefore, the maximum attainment of formate anions in these arrays corresponds to the number of such sandwich complexes rather than the number of porphyrin moieties. The same bonding motif leads to dimers of the composition [(Zn(II)Por/Pc)2·HCOO]−. In these, the formate anion can act as a structural probe, allowing the distinction of isomeric ions with the formate bridging two macrocycles or being attached to a dimer of directly connected macrocycles.
- This article is part of the themed collection: Analyst Recent Open Access Articles


 Please wait while we load your content...
Please wait while we load your content...