“Stealthy” chitosan/mesoporous silica nanoparticle based complex system for tumor-triggered intracellular drug release†
Abstract
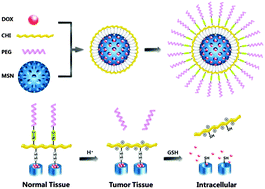
Suitable protection strategies utilized in anticancer drug delivery systems enable carriers to reach their targeted positions and release drugs intracellularly more effectively. In this study, a novel “stealthy” chitosan (CHI)/mesoporous silica nanoparticle (MSN) based complex system, named DOX@MSN-SS-CHI-PEG, was developed for tumor-triggered intracellular drug release. CHI was applied to block the pores of MSNs to prevent premature drug release, whereas mPEG was grafted on the surface of the nanoparticles via a pH-sensitive benzoic imine linker to protect the carriers. As the pH of solid tumor tissues is slightly lower than that of normal tissues, mPEG could leave the nanoparticles to expose positively charged CHI at the surface, which enabled the nanoparticles to enter cancer cells more easily. The MSNs were covered by CHI via redox-sensitive disulfide bonds. As a result, the carriers could release the drug intercellularly to kill cancer cells owing to the high concentration of glutathione (GSH) in the cytosol. In vitro drug release studies at different GSH concentrations proved the redox-sensitivity of DOX@MSN-SS-CHI-PEG. mPEG leaving studies demonstrated that mPEG could leave the nanoparticles effectively at pH 6.0. The cytotoxicity and cell internalization behavior were also investigated in detail. In conclusion, the novel DOX@MSN-SS-CHI-PEG drug delivery system, which was “stealthy” in the physiological environment at pH 7.4 because of the protection of mPEG, was “activated” in weakly acidic tumor tissues to achieve tumor-triggered intracellular drug release; this system has great potential for cancer therapy.
- This article is part of the themed collection: 2016 Journal of Materials Chemistry B Hot Papers

 Please wait while we load your content...
Please wait while we load your content...