Anion-exchange membranes with polycationic alkyl side chains attached via spacer units†
Abstract
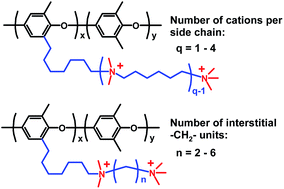
Anion-exchange membrane (AEM) fuel cells are promising electrochemical systems for efficient and environmentally benign energy conversion. However, the development of high-performance fuel cells requires new AEMs tailored for high conductivity and chemical stability. Herein, we present the synthesis and characterization of AEMs with polycationic side chains attached to poly(phenylene oxide) (PPO) via flexible alkyl spacer units. Three series of PPOs were functionalized with side chains to study the influence of the number (n = 2–6) of –CH2– groups in between the quaternary ammonium (QA) cations, the ion exchange capacity (IECs), and the number (q = 1–4) of QA cations per side chain. The polymers were prepared by successively reacting bromoalkylated PPO with different tertiary diaminoalkanes and 1,6-dibromohexane. Evaluation of the alkaline stability by 1H NMR spectroscopy and thermogravimetry demonstrated that solvent cast AEMs with n = 2 and 3 quickly degraded via Hofmann β-elimination in 1 M NaOH at 60 °C. In sharp contrast, no degradation was detected for AEMs with n = 4 and 6 after storage in 1 M NaOH at 90 °C over at least 8 days. At similar IECs, the OH− conductivity of the AEMs increased with n up to n = 4, whereafter a plateau was reached. This may be explained by a polyelectrolyte effect leading to counter ion condensation and incomplete ion dissociation when the QA cations were closer than the Bjerrum length (approx. 7 Å). The conductivity of AEMs with n = 6 and IEC = 1.9 meq. g−1 increased only slightly with the number of QA cations per side chain up to q = 3 but then increased sharply with q = 4 to reach 160 mS cm−1 at 80 °C. The present work demonstrated that a molecular architecture with poly-QA side chains attached via flexible spacer units affords AEMs that combine efficient phase separation, high alkaline stability and OH− conductivity at moderate water uptake, provided that the side chains are properly designed to avoid Hofmann elimination and counter ion condensation.

 Please wait while we load your content...
Please wait while we load your content...