Understanding why replacing I3−/I− by cobalt(ii)/(iii) electrolytes in bis(diimine)copper(i)-based dye-sensitized solar cells improves performance†
Abstract
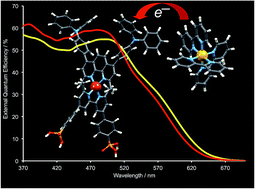
The performances of dye-sensitized solar cells (DSCs) comprising heteroleptic bis(diimine)copper(I) based dyes combined with either [Co(bpy)3]2+/3+, [Co(phen)3]2+/3+ or I3−/I− redox mediators (bpy = 2,2′-bipyridine, phen = 1,10-phenanthroline) have been evaluated. The copper(I) dyes contain the anchoring ligand ((6,6′-dimethyl-[2,2′-bipyridine]-4,4′-diyl)bis(4,1-phenylene))bis(phosphonic acid), 1, and an ancillary ligand (2, 3 or 4) with a 2,9-dimethyl-1,10-phenanthroline metal-binding domain. Ligands 2 and 3 include imidazole 2′-functionalities with 4-(diphenylamino)phenyl (2) or 4-(bis(4-n-butoxy)phenylamino)phenyl (3) domains; in 4, the phen unit is substituted in the 4,7-positions with hole-transporting 4-(diphenylamino)phenyl groups. The photoconversion efficiency, η, of each of [Cu(1)(2)]+, [Cu(1)(3)]+ and [Cu(1)(4)]+ considerably improves by replacing the I3−/I− electrolyte by [Co(bpy)3]2+/3+ or [Co(phen)3]2+/3+, and after a change of electrolyte solvent (MeCN to 3-methoxypropionitrile). Due to the faster charge transfer kinetics and more positive redox potential, the cobalt-based electrolytes are superior to the I3−/I− electrolyte in terms of open-circuit voltage (VOC), short-circuit current (JSC) and η; values of VOC = 594 mV, JSC = 9.58 mA cm−2 and η = 3.69% (relative to η = 7.12% for N719) are achieved for the best performing DSC which contains [Cu(1)(4)]+ and [Co(bpy)3]2+/3+. Corresponding values for [Cu(1)(4)]+ and I3−/I− DSCs are 570–580 mV, 5.98–6.37 mA cm−2 and 2.43–2.62%. Electrochemical impedance spectroscopy (EIS) has been used to study DSCs with [Cu(1)(4)]+ and the three electrolytes. EIS shows that the DSC with I3−/I− has the highest recombination resistance, whereas the [Co(phen)3]2+/3+ electrolyte gives the highest chemical capacitance and VOC and, between [Co(bpy)3]2+/3+ and [Co(phen)3]2+/3+, the higher recombination resistance. The [Co(phen)3]2+/3+ electrolyte exhibits the highest mass transport restrictions which result in a lower JSC and DSC efficiency compared to the [Co(bpy)3]2+/3+ electrolyte.

 Please wait while we load your content...
Please wait while we load your content...