Unconventional structural and morphological transitions of nanosheets, nanoflakes and nanorods of AuNP@MnO2†
Abstract
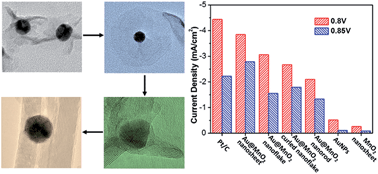
Two-dimensional (2-D) layered inorganic materials with ultra-high surface area and mechanical strength have shown impressive photo-/electro-catalytic activities. We herein report a facile synthetic strategy to grow monodispersed 2-D MnO2 nanosheet on an individual gold nanoparticle (AuNP@MnO2 nanosheet), and demonstrate that the strongly interacted AuNP and MnO2 nanosheet could greatly improve the electrocatalytic activity of the MnOx family for electrocatalytic oxygen reduction reactions (ORRs). AuNP@MnO2 nanosheets were prepared using a hydrothermal reduction of KMnO4 by citrate ligands capped on AuNPs. Because of the metastability of the layered MnO2 nanosheets, we observed unconventional structural and morphological transitions of amorphous MnO2 nanosheets to δ-MnO2 nanoflakes, and eventually to α-MnO2 nanorods under hydrothermal conditions. The layered MnO2 nanosheets underwent a structural expansion to nanoflakes before the curling and re-folding of layered MnO2 nanosheets occurred. The intermediate states and structural transitions via a “layer compression”, for the first time, were experimentally recorded at a single-NP scale using electron microscopy. Moreover, we found the electrocatalytic activity of AuNP@MnO2 nanosheets was enhanced roughly 30–40 times, compared to that of pure MnO2 nanosheets and AuNPs. The strong interaction of metal–oxide interfaces (MnO2 nanosheets and AuNPs) was likely responsible for the improved electrocatalytic activity. This interaction of MnO2 and AuNPs was weakened in the course of hydrothermal treatment where partially positively charged Au+ was reduced at elevated temperatures, accompanying with the decrease of ORR activity. This insight into the effect of topological nanostructures and metal–oxide interactions on the electrocatalytic performance of the MnOx family is believed to illustrate an alternative pathway to develop new efficient electrocatalysts.

 Please wait while we load your content...
Please wait while we load your content...