Carbon nanotube containing Ag catalyst layers for efficient and selective reduction of carbon dioxide†
Abstract
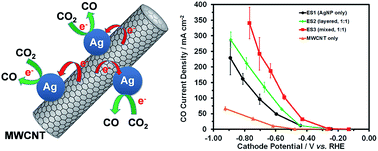
Over the last few decades significant progress has been made in the development of catalysts for efficient and selective electroreduction of CO2. These improvements in catalyst performance have been of the extent that identifying electrodes of optimum structure and composition has become key to further improve throughput levels in the electrolysis of CO2 to CO. Here we report on a simple one-step method to incorporate multi-walled carbon nanotubes (MWCNT) in the catalyst layer to form gas diffusion electrodes with different structures: (i) a “mixed” catalyst layer in which the Ag nanoparticle catalyst and MWCNTs are homogeneously distributed; and (ii) a “layered” catalyst layer comprised of a layer of MWCNTs covered with a layer of Ag catalyst. Both approaches improve performance in the electroreduction of CO2 compared to electrodes that lack MWCNTs. The “mixed” layer performed best: an electrolyzer operated at a cell potential of −3 V using 1 M KOH as the electrolyte yielded unprecedented high levels of CO production of up to 350 mA cm−2 at high faradaic efficiency (>95% selective for CO) and an energy efficiency of 45% under the same condition. Electrochemical impedance spectroscopy measurements indicate that the observed differences in electrode performance can be attributed to a lower charge transfer resistance in the “mixed” catalyst layer. This study shows that a simple optimization of electrode structure and composition, i.e. incorporation of MWCNTs in the catalyst layer of a GDE, has a profound beneficial effect on their performance in electrocatalytic conversion of CO2, while allowing for a lower precious metal catalyst loading with improved performance.


 Please wait while we load your content...
Please wait while we load your content...