Kinetic analysis of photoelectrochemical water oxidation by mesostructured Co-Pi/α-Fe2O3 photoanodes†
Abstract
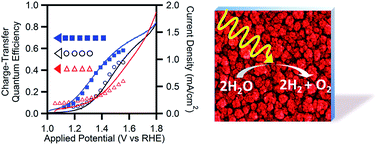
Solar water splitting using catalyst-modified semiconductor photoelectrodes is a promising approach to harvesting and storing solar energy. Prior studies have demonstrated that modification of α-Fe2O3 photoanodes with the water-oxidation electrocatalyst Co-Pi enhances photon-to-current conversion efficiencies, particularly at less positive potentials, but the mechanism underlying this enhancement remains poorly understood. Different experimental techniques have suggested very different interpretations of the microscopic origins of this improvement. Here, we report results from photoelectrochemical and impedance measurements aimed at understanding the Co-Pi/α-Fe2O3 interface of mesostructured composite photoanodes. Contrary to expectations, these measurements reveal that α-Fe2O3 water-oxidation kinetics actually slow upon deposition of Co-Pi, but electron–hole recombination slows even more, resulting in a net enhancement of water-oxidation quantum efficiency. The negative shift in the J–V curve caused by Co-Pi deposition is found to result from the introduction of an alternative pathway for water oxidation catalyzed by Co-Pi, which allows the composite photoanode to avoid positive charge accumulation at the α-Fe2O3 surface. We detail the role of Co-Pi thickness optimization in balancing the slower recombination against the slower water oxidation kinetics to achieve the lowest water-oxidation onset potential. These results provide new insights into the microscopic properties of the catalyst/semiconductor interface in Co-Pi/α-Fe2O3 composite solar water-splitting photoanodes.
- This article is part of the themed collection: Water splitting and photocatalysis

 Please wait while we load your content...
Please wait while we load your content...