Using depletion to control colloidal crystal assemblies of hard cuboctahedra†
Abstract
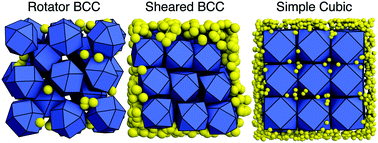
Depletion interactions arise from entropic forces, and their ability to induce aggregation and even ordering of colloidal particles through self-assembly is well established, especially for spherical colloids. We vary the size and concentration of penetrable hard sphere depletants in a system of cuboctahedra, and we show how depletion changes the preferential facet alignment of the colloids and thereby selects different crystal structures. Moreover, we explain the cuboctahedra phase behavior using perturbative free energy calculations. We find that cuboctahedra can form a stable simple cubic phase, and, remarkably, that the stability of this phase can be rationalized only by considering the effects of both the colloid and depletant entropy. We corroborate our results by analyzing how the depletant concentration and size affect the emergent directional entropic forces and hence the effective particle shape. We propose the use of depletants as a means of easily changing the effective shape of self-assembling anisotropic colloids.

 Please wait while we load your content...
Please wait while we load your content...