Catalytic radical reduction in aqueous solution via oxidation of biologically-relevant alcohols†
Abstract
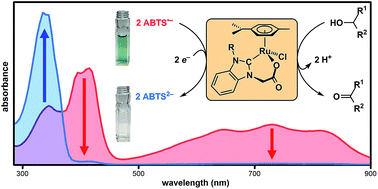
Metalloenzymes that normally perform catalytic antioxidant or radical-degrading functions, as well as small-molecule complexes that mimic them, can also exert pro-oxidant or radical-forming effects depending on the identity of the terminal reductant. Because nitroxyl radicals function as redox active cocatalysts in the aerobic oxidation of alcohols, we hypothesized that catalytic radical reduction could be achieved via the oxidation of biologically-relevant alcohols. Herein we report an organoruthenium complex (Ru1) that catalyzed reduction of 2,2′-azino-bis(3-ethylbenzo-thiazoline-6-sulfonate) radical monoanion (ABTS˙−) to ABTS2− in phosphate buffered saline (pH 7.4) using MeOH, EtOH, i-PrOH, serine, threonine, glucose, arabinose, methyl lactate or dimethyl malate as the terminal reductant. Replacing either the C–H or O–H groups of a –CHOH– moiety resulted in the loss of ABTS˙− reducing ability. Moreover, in conjunction with an alcohol terminal reductant, Ru1 was able to inhibit the oxidation of ABTS2− by H2O2 and horseradish peroxidase, even after multiple successive challenges with excess H2O2 or ABTS˙−. Collectively, these results demonstrate that Ru1 inhibits the oxidative formation of and catalyzes the reduction of radicals in aqueous solution via oxidation of biologically-relevant alcohols.

 Please wait while we load your content...
Please wait while we load your content...