On the agent role of Mn2+ in redirecting the synthesis of Zn(OH)2 towards nano-ZnO with variable morphology†
Abstract
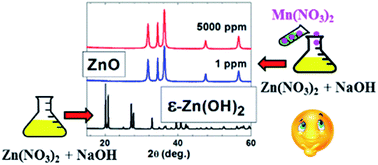
One of the simplest routes to prepare polycrystalline Zn(OH)2 is by coprecipitation, with zinc nitrate as a cation source. However, the addition of even minute amounts of manganese nitrate to the precursors used to prepare pure Zn(OH)2 results in Mn2+ doped nanostructured ZnO. The comparison with other Mn2+ doped metal hydroxides prepared by the same coprecipitation method, involving metal nitrates precursors, shows that this behavior is unique, pertaining only to Zn(OH)2. A systematic study of the samples prepared without and with variable amounts of Mn2+ ions, in the 1 to 5000 ppm nominal concentrations range showed that the re-routing of the reaction takes place even for the lowest nominal dopant concentration of 1 ppm. According to X-ray diffraction, transmission electron microscopy and Fourier transform infrared spectroscopy investigations, both crystallite size and morphology of the resulting nanostructured ZnO samples varied with the Mn2+ nominal concentration. Moreover, quantitative electron paramagnetic resonance investigations showed that the incorporation rate of the Mn2+ ions at different sites in the nanostructured ZnO depended on the nominal Mn2+ concentration. The results are discussed in terms of the coordination properties of the Mn2+ and Zn2+ ions and the nature of the reaction precursors.


 Please wait while we load your content...
Please wait while we load your content...