Green Schiff's bases as corrosion inhibitors for mild steel in 1 M HCl solution: experimental and theoretical approach
Abstract
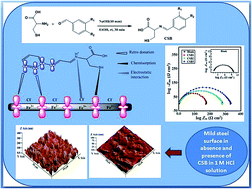
Three cysteine based Schiff's bases namely 3-mercapto-2-((4-methoxybenzylidene)amino)propanoic acid (CSB-1), 2-((4-hydroxy-3-methoxybenzylidene)amino)-3-mercaptopropanoic acid (CSB-2) and 3-mercapto-2-(((E)-3-phenylallylidene)amino)propanoic acid (CSB-3) were synthesized and their corrosion inhibition properties on mild steel in 1 M HCl solution were evaluated using weight loss, electrochemical study and quantum chemical calculations. The result showed that CSB-3 is the best among the three studied inhibitors and showed the efficiency of 97.3% at 200 ppm concentration. The adsorption of inhibitors was found to be both physisorption and chemisorption and followed Langmuir's adsorption isotherm. SEM and AFM study also confirmed the formation of protective films of the inhibitors on mild steel surface. Potentiodynamic polarization studies revealed that all CSBs were of mixed type and predominantly behaved as cathodic inhibitors. EIS study showed that they inhibit corrosion by increasing the charge transfer resistance between metal–solution interfaces. Quantum chemical calculation parameters such as EHOMO, ELUMO, ΔE, global hardness and softness, electronegativity and fraction of electron transfer (ΔN) were calculated using DFT method to correlate the electronic properties with the adsorption behavior of studied Schiff bases.

 Please wait while we load your content...
Please wait while we load your content...