Highly efficient continuous-flow oxidative coupling of amines using promising nanoscale CeO2–M/SiO2 (M = MoO3 and WO3) solid acid catalysts†
Abstract
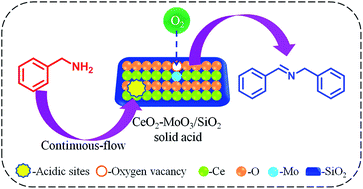
The development of promising solid acid catalysts alternative to hazardous liquid acids is essential towards a sustainable chemical industry. This work reports the synthesis of nanostructured CeO2–MoO3/SiO2 and CeO2–WO3/SiO2 solid acids, along with CeO2–MoO3, CeO2–WO3 and CeO2 for continuous-flow oxidative coupling of benzylamine using O2 as a green oxidant. A systematic physicochemical characterization has been undertaken using XRD, Raman, N2 adsorption–desorption, TEM, NH3-TPD, and XPS techniques. It was found that the dispersion of CeO2–MoO3 and CeO2–WO3 species on the SiO2 support leads to remarkable structural and acidic properties, due to the synergetic effect of the respective components. TEM analysis reveals the presence of highly dispersed WO3 (0.8–1.2 nm) and MoO3 (0.8–1 nm) nanoparticles in the synthesized catalysts. Among the various catalysts developed, the CeO2–MoO3/SiO2 sample exhibited higher BET surface area (248 m2 g−1), abundant oxygen vacancy defects, and large amounts of strong acidic sites. Owing to improved properties, the CeO2–MoO3/SiO2 solid-acid showed a superior catalytic performance in the continuous-flow oxidative coupling of benzylamine: the obtained benzylamine conversions for 1 h are ∼11.8, 55, 70, 76, and 96%, respectively, for CeO2, CeO2–WO3, CeO2–WO3/SiO2, CeO2–MoO3, and CeO2–MoO3/SiO2 catalysts. Importantly, the CeO2–MoO3/SiO2 solid acid exhibited a remarkable steady performance in terms of benzylamine conversion (∼88–96%) and selectivity of N-benzylbenzaldimine product (∼96–97.8%) up to 6 h. The outstanding catalytic performance of CeO2–MoO3/SiO2 solid acid coupled with the application of continuous-flow synthesis, economical benefits of the respective oxides, and eco-friendly oxidant is expected to bring new opportunities in the design of industrially-favourable chemical processes.

 Please wait while we load your content...
Please wait while we load your content...