Self-encapsulation of a drug-containing ionic liquid into mesoporous silica monoliths or nanoparticles by a sol–gel process†
Abstract
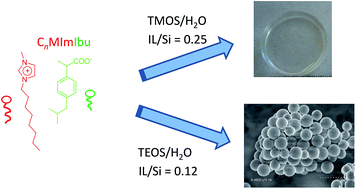
The direct sol–gel encapsulation of an ionic liquid comprising a long alkyl chain imidazolium cation (1-methyl-3-octyl-imidazolium) and ibuprofenate anion, an active pharmaceutical ingredient, has been investigated. Silica ionogels either in the form of centimeter sized monoliths or nanometer sized particles were obtained depending on the conditions used. In both cases, the ionic liquid acted as a templating agent and a catalyst, promoting its own encapsulation. Monolithic ionogels were prepared by a simple hydrolytic method using tetramethoxysilane as the silica source. High pore volumes were measured after removal of the ionic liquid, in relation with the bulkiness of the ions. Typically, surface areas and pore volumes as high as 812 m2 g−1 and 1.25 cm3 g−1 were obtained using a low ionic liquid to silica ratio equal to 0.25, but still representing a high Ibuprofen loading equal to 46 wt%. At the same time, the ibuprofenate anion acted as a catalyst for the sol–gel reaction and a relatively high condensation degree was obtained (88%). Moreover, it was possible to benefit from the fact that the ionic liquid self-organized into micelles in aqueous solutions to incorporate it into mesoporous silica nanospheres in the absence of any organic solvent or other additives using tetraethoxysilane as the silica source. The mechanism involved was evidenced by transmission electron microscopy (TEM). Spherical silicate micelles of 30–40 nm were first formed, followed by their aggregation into spherical nanoparticles. Effects of time, temperature, and ionic liquid concentration on the formation of these silica nanospheres were also investigated.

 Please wait while we load your content...
Please wait while we load your content...