Effect of precursor on the catalytic properties of Ni2P/SiO2 in methyl palmitate hydrodeoxygenation†
Abstract
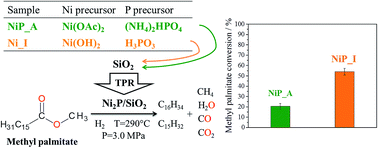
The effect of phosphorus precursor on the physicochemical and catalytic properties of silica-supported nickel phosphide catalysts in the hydrodeoxygenation (HDO) of aliphatic model compound methyl palmitate (C15H31COOCH3) has been considered. Nickel acetate (Ni(OAc)2) and diammonium hydrogen phosphate ((NH4)2HPO4) (phosphate precursor) or nickel hydroxide (Ni(OH)2) and phosphorous acid (H3PO3) (phosphite precursor) have been used for the catalyst preparation by incipient wetness impregnation of SiO2 with an aqueous solution of the precursors, followed by temperature-programmed reduction in hydrogen flow. Chemical analysis (ICP-AES), H2-TPR, NH3-TPD, 31P MAS NMR, XRD, and TEM have been employed for the characterization of the catalysts. The optimal reduction parameters of the silica-supported nickel phosphide catalysts have been found for the phosphate and phosphite precursors in terms of their activity in methyl palmitate HDO. The Ni2P/SiO2 catalysts prepared by the phosphite method have shown higher catalytic activity in comparison with the Ni2P/SiO2 catalysts prepared by the phosphate method. The activity has been shown to depend on the catalyst handling after reduction: in situ reduced catalysts demonstrate higher conversion of methyl palmitate than the samples exposed to the reduction, passivation and re-reduction steps.


 Please wait while we load your content...
Please wait while we load your content...