An investigation on the influence of support type for Ni catalysed vapour phase hydrogenation of aqueous levulinic acid to γ-valerolactone†
Abstract
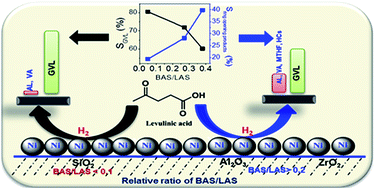
Ni (20 wt%) supported on SiO2, γ-Al2O3 and ZrO2 catalysts was examined for hydrogenation of aqueous levulinic acid (LA) to γ-valerolactone (GVL) at 270 °C and ambient pressure. The band intensities of Brønsted (BAS: 1540 cm−1) and Lewis acid sites (LAS: 1450 cm−1) estimated by pyridine adsorbed DRIFT spectra revealed a lower ratio of BAS/LAS over the Ni/SiO2 catalyst than over the Ni/ZrO2 and Ni/γ-Al2O3 catalysts. The rate of angelica lactone (AL) formation was lower than the rate of AL hydrogenation over the Ni/SiO2 catalyst. The poisoning and regeneration of the Ni/SiO2 catalyst using pyridine and 2,6-dimethylpyridine demonstrated that Lewis acid sites influenced the conversion of LA to AL and subsequent hydrogenation of AL to GVL occurred on surface Ni sites. In contrast, Brønsted acid sites were responsible for the ring opening of GVL to valeric acid (VA). Kinetic data emphasized that the hydrogenation activity and product distribution were dependent on the type of acid site, and the Ni sites in close proximity to Brønsted acid sites are prone to hydrogenolysis of GVL to valeric acid and hydrocarbons.

 Please wait while we load your content...
Please wait while we load your content...