Comparison of hydrogen, halogen, and tetrel bonds in the complexes of HArF with YH3X (X = halogen, Y = C and Si)†
Abstract
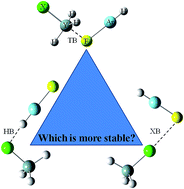
Ab initio MP2/aug-cc-pVTZ calculations were performed in order to find equilibrium structures with Y⋯F tetrel bonds, X⋯H hydrogen bonds or X⋯F halogen bonds on the potential energy surfaces of the complexes formed between HArF and YH3X (X = halogen, Y = C and Si). For the CH3X complexes, the hydrogen-bonded complex is the most stable, while the tetrel-bonded complex is the most stable of the SiH3X complexes. The H–Ar stretch vibration exhibits a red shift for the hydrogen bond but a blue shift for the tetrel and halogen bonds. The hydrogen bonds in the CH3X and SiH3X complexes, as well as the tetrel bonds in the SiH3X complexes, are governed by a combination of electrostatic and polarization energies, exhibiting partially covalent character with negative energy densities and a substantial amount of charge transfer.
- This article is part of the themed collection: 1st International Conference on Noncovalent Interactions

 Please wait while we load your content...
Please wait while we load your content...