Electrochemical oxidation of guaiacol to increase its biodegradability or just remove COD in terms of anodes and electrolytes†
Abstract
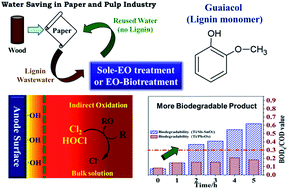
To assess the role of electrochemical oxidation as a single mineralization technique or a pretreatment (for biotreatment) of lignin-containing wastewater, Ti/Sb-SnO2 and Ti/Pb3O4 were used as comparable anode materials to degrade the lignin monomer guaiacol. The electrode catalytic activity was examined by cyclic voltammetry. Then the guaiacol degradation was analyzed. Finally, the main intermediates and products were identified. The results show that Ti/Sb-SnO2 is non-selective in treating guaiacol and its intermediates while Ti/Pb3O4 has a weaker ability in degrading the intermediates. A chlorine-containing media is suitable to perform total organic removal with a high rate, but the active chlorine and organochlorine may ruin the follow-up biological treatment. Electrochemical oxidation (EO) pretreatment could save 50–70% electric energy and reduce the burden of subsequent biotreatment. Quinones and dimers are important intermediates that could be used to distinguish the capability of anodes and the function of Cl−.

 Please wait while we load your content...
Please wait while we load your content...