ent-Abietane diterpenoids with anti-neuroinflammatory activity from the rare Chloranthaceae plant Chloranthus oldhamii†
Abstract
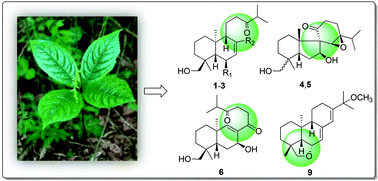
Twelve new ent-abietane diterpenoids, chlorabietins A–L (1–12), were isolated from the roots of Chloranthus oldhamii. Their structures and absolute configurations were determined by extensive spectroscopic analyses, X-ray diffraction, and experimental/calculated electronic circular dichroism (ECD) spectroscopy. Among the new isolates, chlorabietins D (4) and E (5) are the first two naturally occurring 8-spiro-fused 9,10-seco-ent-abietanes containing an unexpected cis-fused A/B ring system. Chlorabietin F (6) is a rare chinane-type diterpenoid featuring a hitherto unknown C-ring cleavage between C-13 and C-14, which might be derived from a common precursor of the above spiro-diterpenoid epimers 4 and 5, and their biosynthetic relationships are briefly discussed. Meanwhile, chlorabietin I (9) is the first representative of the abietane-type diterpenoids possessing a tetrahydrofurano function bridging C-6 and C-19. Chlorabietins B (2), C (3), F (6), and G (7) showed anti-neuroinflammatory effects by inhibiting the nitric oxide (NO) production in lipopolysaccharide (LPS)-activated murine BV-2 microglial cells, with IC50 values ranging from 16.4 to 33.8 μM.

 Please wait while we load your content...
Please wait while we load your content...