Compact quantum dot–antibody conjugates for FRET immunoassays with subnanomolar detection limits†
Abstract
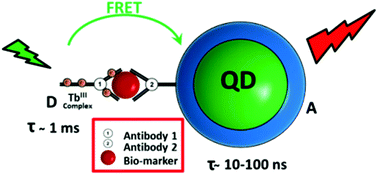
A novel two-step approach for quantum dot (QD) functionalization and bioconjugation is presented, which yields ultra-compact, stable, and highly luminescent antibody–QD conjugates suitable for use in FRET immunoassays. Hydrophobic InPZnS/ZnSe/ZnS (emission wavelength: 530 nm), CdSe/ZnS (605 nm), and CdSeTe/ZnS (705 nm) QDs were surface functionalized with zwitterionic penicillamine, enabling aqueous phase transfer under conservation of the photoluminescence properties. Post-functionalization with a heterobifunctional crosslinker, containing a lipoic acid group and a maleimide function, enabled the subsequent coupling to sulfhydryl groups of proteins. This was demonstrated by QD conjugation with fragmented antibodies (F(ab)). The obtained F(ab)–QD conjugates range among the smallest antibody-functionalized nanoprobes ever reported, with a hydrodynamic diameter <13 nm, PL quantum yield up to 66% at 705 nm, and colloidal stability of several months in various buffers. They were applied as FRET acceptors in homogeneous, time-gated immunoassays using Tb-antibodies as FRET donors, both coupled by an immunological sandwich complex between the two antibodies and a PSA (prostate specific antigen) biomarker. The advantages of the compact surface coating for FRET could be demonstrated by an 6.2 and 2.5 fold improvement of the limit of detection (LOD) for PSA compared to commercially available hydrophilic QDs emitting at 605 and 705 nm, respectively. While the commercial QDs contain identical inorganic cores responsible for their fluorescence, they are coated with a comparably thick amphiphilic polymer layer leading to much larger hydrodynamic diameters (>26 nm without biomolecules). The LODs of 0.8 and 3.7 ng mL−1 obtained in 50 μL serum samples are below the clinical cut-off level of PSA (4 ng mL−1) and demonstrate their direct applicability in clinical diagnostics.

 Please wait while we load your content...
Please wait while we load your content...