Selective modification of the metal coordination environment in heavy alkaline–earth iodide complexes†
Abstract
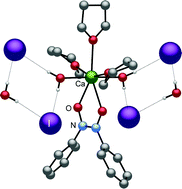
A series of heavy alkaline–earth (Ae) iodide coordination compounds is reported that contains various neutral donor ligands, including triphenylphosphine oxide (OPPh3), tri-n-propylphosphine oxide (OPPr3), tetraethyl urea (TEU), dimethylpropylene urea (DMPU), and the nitrosobenzene dimer ([ONPh]2). Phosphine oxides can displace iodide from calcium or barium in coordination complexes, sometimes displaying cooperative binding and/or generating cationic species of the general formula [AeIm(OPR3)6−(m+p)(THF)p](2−m)+. In particular, despite the nonstoichiometric ratio of the reagents, 2 equiv. of OPPh3 react with CaI2 in THF to generate CaI2(OPPh3)3(THF) (1), although the use of 4 equiv. will produce the expected CaI2(OPPh3)4 (2). With CaI2 and 5 equiv. of tri(n-propyl)phosphine oxide, the cationic species [CaI(OPPr3)5]I (3) is formed. With 4 equiv. of OPPh3 and BaI2 in THF, the cation [BaI(OPPh3)5]I (4) is generated. For comparative purposes, the ureas tetraethylurea (TEU) and N,N′-dimethylpropylene urea (DMPU) were used to form the complexes CaI2(TEU)4 (5) and CaI2(DMPU)6 (6). Nitrosobenzene reacts with CaI2 in THF to form CaI2(O2N2Ph2)2(THF)2, which in the presence of trace amounts of water is converted to the hydrogen-bonded [Ca(O2N2Ph2)(H2O)2(THF)3]I2 (7). Crystal structures are reported for 3–5 and 7; the latter is the first for the nitrosobenzene dimer in a main group complex.


 Please wait while we load your content...
Please wait while we load your content...