Dispersion forces acting between silica particles across water: influence of nanoscale roughness†
Abstract
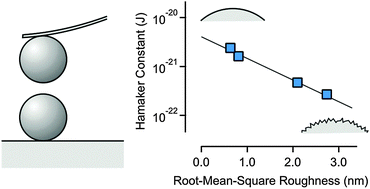
Force profiles between pairs of silica particles in concentrated aqueous solutions of a monovalent salt are measured using atomic force microscopy (AFM). Under such conditions, the double layer forces are negligible, and the profiles are dominated by van der Waals dispersion forces at larger distances. Heat treatment of the particles strongly influences the strength of dispersion forces. The dispersion force between the particles heated at 1200 °C was strongly attractive, and was characterized by a Hamaker constant of 2.4 × 10−21 J. This value is in good agreement with the current best theoretical estimate of the Hamaker constant for silica across water. For untreated particles, however, the dispersion force is much weaker and the Hamaker constant is 7 × 10−23 J. The Hamaker constant can be continuously tuned by adjusting the heating temperature between 1000 and 1200 °C. Such substantial variations of the Hamaker constant are caused by moderate differences in surface roughness on the nanoscale. The root mean square (RMS) of the roughness correlates inversely with the Hamaker constant, whereby the particles treated at 1200 °C have an RMS value of 0.63 nm, while the untreated particles have an RMS value of 2.5 nm. Other effects that could influence the Hamaker constant, such as changes in the degree of crystallinity, porosity, and shape of the particles, could be excluded.

 Please wait while we load your content...
Please wait while we load your content...