Precise measurement of Cr isotope ratios using a highly sensitive Nb2O5 emitter by thermal ionization mass spectrometry and an improved procedure for separating Cr from geological materials
Abstract
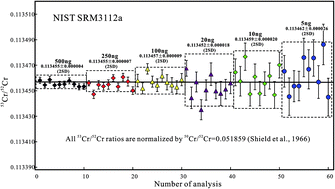
Chromium isotopes have important applications in cosmochemistry, environmental science, geochemistry, and paleoceanography studies. In previous investigations, thermal ionization mass spectrometry (TIMS) showed excellent precision for Cr isotopic ratios analysis. However, relative large sample sizes (200–2000 ng) were required for TIMS measurements and therefore hampered its wider application in geoscience and environmental science. In this study, a highly sensitive Nb2O5 emitter is employed for the first time to measure Cr isotopes using TIMS. This Nb2O5 emitter produces at least a 10-fold enhancement in the sensitivity and can not only significantly reduce the sample size, but also obtain good external precision. In this study, an external precision of ±0.23‰ (2 SD, n = 10) on δ53Cr was obtained on a 5 ng sample of NIST SRM 3112a standard. In addition, an improved two-column chemical separation procedure was devised in order to separate Cr from complex geological materials. Compared to the classical anion resin purification method, the procedural blank and severe inhibition effect from SO42− is significantly reduced by carefully optimizing and evaluating the dosage of (NH4)2S2O8 oxidant. This method has been verified using one dolomite, three basalt and one peridotite reference materials. A range of analytical tests demonstrates that it is possible to attain internal precisions (2 SE) of ±0.017 to 0.037‰ on a single 30 min analysis. Long-term reproducibility (2 SD) for 53Cr/52Cr is 0.113454 ± 0.000004 (∼0.035‰) obtained by replicate analyses (n = 105) of NIST SRM 3112a over a 6 month period. Replicate digestions and analyses of the basalt standard BIR-1 (δ53Cr = −0.110 ± 0.048‰, 2 SD) and the carbonate standard JDo-1 (δ53Cr = 1.710 ± 0.054‰, 2 SD) demonstrate that good external reproducibility is obtainable on natural samples.

 Please wait while we load your content...
Please wait while we load your content...