Chemical speciation via X-ray emission spectroscopy in the tender X-ray range
Abstract
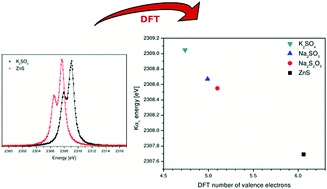
Kα X-ray emission spectra from a series of phosphorus, sulfur, and chlorine containing compounds covering the full range of oxidation states were measured employing high energy resolution proton induced X-ray emission (PIXE) spectroscopy in the tender X-ray range. Measurements were accompanied by quantum chemistry calculations based on density functional theory (DFT). Clear energy shifts of the Kα lines were measured in correlation with the formal oxidation state for all three elements. This correlation was improved even further using the effective charge based on the DFT calculated valence electron population. Finally, it is demonstrated that the oxidation state analysis based on the Kα energy shifts can be used also for a quantitative analysis of the proportion of separate low-Z species in mixed valence systems.

 Please wait while we load your content...
Please wait while we load your content...