Compositional and structural feedstock requirements of a liquid phase cellulose-to-naphtha process in a carbon- and hydrogen-neutral biorefinery context†
Abstract
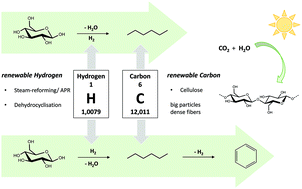
Processing raw (ligno)cellulosic feedstock into renewable light naphtha alkanes could lead to a gradual replacement of fossil feedstock for the production of chemicals, materials and fuels. The production of drop-in alkanes is a preferable short term strategy because of its practical implementation and integration in existing infrastructure and processes. A handful of promising cellulose-to-alkane biorefinery initiatives were recently reported, both processing in gas and liquid phase. This contribution presents a detailed study of the two-liquid phase hydrodeoxygenation of cellulose to n-hexane under relatively mild circumstances, proceeding through the recently communicated HMF route, in presence of a soluble acid and Ru/C metal catalyst. Two main points were addressed here: (i) the importance (or not) of the lignocellulose pretreatment and purification to the alkane yield, and (ii) the renewability of the consumed hydrogen in the process. A systematic study of the effect of cellulose purity, crystallinity, degree of polymerization and particle size (surface area) on the light naphtha yield was performed to tackle the first part. As fibrous cellulose with large particles was the most favourable feedstock with regard to alkane yield and as the presence of hemicellulose and lignin impurities had no effect on the cellulose-to-naphtha conversion, costly mechanical and purification steps are redundant to the process, in contrast to their notable importance in other cellulose valorisation processes (e.g. to glucose, sorbitol, isosorbide and acids). The second point regarding sustainable hydrogen supply is discussed in detail by calculating hydrogen and carbon mass and energy balances of the chemical conversions, assuming selected scenarios among others to recuperate the hydrogen by steam-reforming of waste streams (like gaseous C<6 hydrocarbons and aqueous polyol fractions) and (partial) aromatization of the C6 fraction into benzene. The study shows potential to integrate the liquid phase cellulose-to-naptha (LPCtoN) technology into a self-sufficient biorefinery, in which the chemical processes may run without consumption of external (non-renewable) hydrogen, carbon and energy, except for solar light.

 Please wait while we load your content...
Please wait while we load your content...