Chemo-enzymatic preparation and characterization of renewable oligomers with bisguaiacol moieties: promising sustainable antiradical/antioxidant additives†
Abstract
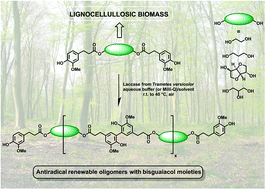
The synthesis, structural characterization and properties of new bio-based oligomers with bisguaiacol-type moieties esterified by diverse aliphatic linkers are described. These oligomers, produced by oxidative oligomerization of renewable dihydroferulic acid-based bisphenols with commercially available Trametes versicolor laccase, are proposed as promising sustainable antiradical/antioxidant additives for polymers. This environmentally friendly biocatalyzed process is performed under very mild conditions in open vessels and aqueous solution at room temperature. Modifications of the reaction conditions (use of an organic co-solvent up to 80% v/v and increase of the reaction temperature up to 60 °C) revealed that the polymerization degree of the oligomers may be controlled by adjusting the nature and the ratio of the co-solvent, the reaction time and the reaction temperature. Thermal analyses (TGA and DSC) demonstrated that these phenolic oligomers exhibit high thermal stability and that their Td 5% and Tg can be easily tailored by playing with both the structure of the bisphenol and the degree of polymerization. Similarly, these phenolic oligomers exhibit tunable potent antiradical/antioxidant activity as shown by DPPH analyses. These aliphatic–aromatic oligomers with bisguaiacol-type moieties are thus promising as easily accessible, eco-friendly antiradical/antioxidant additives for the stabilization of polymers in packaging and other applications.

 Please wait while we load your content...
Please wait while we load your content...