A large number of low coordinated atoms in boron nitride for outstanding adsorptive desulfurization performance†
Abstract
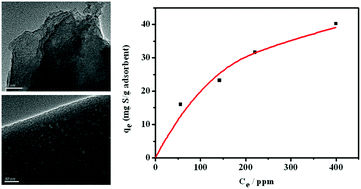
h-BN has been demonstrated to be able to exhibit adsorptive desulfurization from fuel. In order to further optimize the adsorption capacities to meet the potential industrial applications, tuning the nanostructure of BN was taken into account. In this work, we demonstrated that cyanamide, dicyandiamide, and melamine as different nitrogen precursors for synthesizing BN could tune the BN nanoarchitectures. The high performance BN prepared with melamine presented a ribbon-like structure which was assembled with porous nanosheets. This kind of nanoarchitecture with exposed BN sharp edges and a porous structure can be constructed on the BN surface. The large number of low coordinated atoms at the exposed sharp edges and along the edges of the pores could build powerful interaction with sulfide, which was believed to be responsible for the advanced adsorption capacity. The prepared BN with melamine as nitrogen precursors displayed remarkable adsorption performance for DBT (40.2 mg S per g adsorbent for 500 ppm sulfur model oil and 57.5 mg S per g adsorbent according to the Langmuir isotherm model). To the best of our knowledge, it is the highest adsorption capacities reported so far for the adsorptive desulfurization. It is also noteworthy to mention that even for refractory sulfur compound 4,6-DMDBT, the prepared BN still showed high adsorption performance.

 Please wait while we load your content...
Please wait while we load your content...