Triplet state dissolved organic matter in aquatic photochemistry: reaction mechanisms, substrate scope, and photophysical properties
Abstract
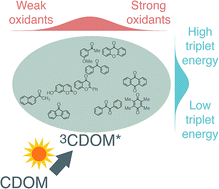
Excited triplet states of chromophoric dissolved organic matter (3CDOM*) play a major role among the reactive intermediates produced upon absorption of sunlight by surface waters. After more than two decades of research on the aquatic photochemistry of 3CDOM*, the need for improving the knowledge about the photophysical and photochemical properties of these elusive reactive species remains considerable. This critical review examines the efforts to date to characterize 3CDOM*. Information on 3CDOM* relies mainly on the use of probe compounds because of the difficulties associated with directly observing 3CDOM* using transient spectroscopic methods. Singlet molecular oxygen (1O2), which is a product of the reaction between 3CDOM* and dissolved oxygen, is probably the simplest indicator that can be used to estimate steady-state concentrations of 3CDOM*. There are two major modes of reaction of 3CDOM* with substrates, namely triplet energy transfer or oxidation (via electron transfer, proton-coupled electron transfer or related mechanisms). Organic molecules, including several environmental contaminants, that are susceptible to degradation by these two different reaction modes are reviewed. It is proposed that through the use of appropriate sets of probe compounds and model photosensitizers an improved estimation of the distribution of triplet energies and one-electron reduction potentials of 3CDOM* can be achieved.
- This article is part of the themed collection: Natural Organic Matter


 Please wait while we load your content...
Please wait while we load your content...