Comparison of heterogenized molecular and heterogeneous oxide catalysts for photoelectrochemical water oxidation†
Abstract
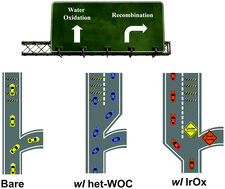
Photoelectrochemical (PEC) reactions, such as water splitting, promise a direct route for solar-to-chemical energy conversion. Successful implementations of these reactions often require the combination of catalysts with photoelectrodes. How these catalysts improve the performance of photoelectrodes, however, is not well understood, making it difficult to further improve these systems for practical applications. Here, we present a systematic study that directly compares two water-oxidation catalysts (WOCs) on a hematite (α-Fe2O3)-based PEC system. We observe that when a thin layer of a heterogenized molecular Ir catalyst (het-WOC) is applied to a hematite photoanode, the system's performance is improved primarily due to improved charge transfer (>2 fold), while the surface recombination rate remains unchanged. In stark contrast, heterogeneous oxide catalysts (IrOx) improve the PEC performance of hematite by significantly reducing the surface recombination rate. These results suggest that the het-WOC provides additional charge-transfer pathways across the Fe2O3|H2O interface, while IrOx and similar bulk metal-oxide catalysts replace the Fe2O3|H2O interface with a fundamentally different one.

 Please wait while we load your content...
Please wait while we load your content...