Bioisosteric ferrocenyl-containing quinolines with antiplasmodial and antitrichomonal properties†
Abstract
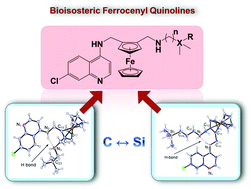
Bioisosteric ferrocenyl-containing quinolines and ferrocenylamines containing organosilanes and their carbon analogues, were prepared and fully characterised. The molecular structures of two ferrocenyl-containing quinolines, determined using single-crystal X-ray diffraction, revealed that the compounds crystallise in a folded conformation. The compounds were screened for their antiplasmodial activity against the chloroquine-sensitive (NF54) and CQ-resistant (Dd2) strains of P. falciparum, as well as for their cytotoxicity against Chinese Hamster Ovarian (CHO) cells. The ferrocenyl-containing quinolines displayed activities in the low nanomolar range (6–36 nM), and showed selectivity towards parasites. β-Haematin inhibition assays suggest that the compounds may in part act via the inhibition of haemozoin formation, while microsomal metabolic stability studies reveal that the ferrocenyl-containing quinolines are rapidly metabolised in liver microsomes. Further, antitrichomonal screening against the metronidazole-sensitive (G3) strain of the mucosal pathogen T. vaginalis revealed that the quinoline-based compounds displayed superior parasite growth inhibition when compared to the ferrocenylamines. The library was also tested E. coli and on Lactobacilli spp. found as part of the normal flora of the human microbiome and no effect on growth in vitro was observed, supporting the observation that these compounds are specific for eukaryotic pathogens.

 Please wait while we load your content...
Please wait while we load your content...