Understanding and tuning the properties of redox-accumulating manganese helicates†
Abstract
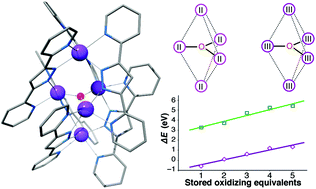
The pentanuclear Mn complex [Mn5(μ3-O)(bpp)6]n+ can access six consecutive total oxidation levels from Mn(II)5 to Mn(III)5. The electronic structure and redox behavior of this cluster are studied computationally and the results are compared with experimental data. The tunability of the redox potential span and of the absolute potential position is explored through systematic modifications of the 3,5-bis(pyridin-2-yl)-pyrazole (Hbpp) ligand. By substitutions with electron-donating and electron-withdrawing groups, the position of the redox events can be shifted by ca. 1 eV, however no ligand modification is predicted to alter the total redox span. By changing the pyridyl groups to benzimidazole groups, yielding the 3,5-bis(benzimidazol-2-yl)pyrazole (H3bbp) ligand, it is predicted that higher oxidation states can be stabilized, from Mn(III)4Mn(IV) up to Mn(IV)5. In this system, the redox span for the same number of redox equivalents accumulated is slightly smaller than that in the original system. The manganese system is compared with its pentanuclear iron analogue that was recently reported to be catalytically active in oxygen evolution (Okamura et al., Nature, 2016, 530, 465). The electronic and structural requirements for utilization of the stored oxidizing equivalents in water oxidation are discussed.


 Please wait while we load your content...
Please wait while we load your content...