Chiral amino-phosphine and amido-phosphine complexes of Ir and Mg. Catalytic applications in olefin hydroamination†
Abstract
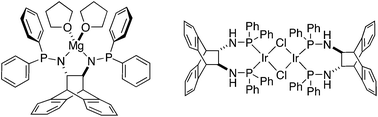
The reactions of rac- and (S,S)-trans-9,10-dihydro-9,10-ethanoanthracene-11,12-diamine (ANDEN) with PClPh2 in the presence of NEt3 yield the chiral amino-phosphine ligands rac-6 and (S,S)-6, respectively, on multi-gram scales. Both forms of 6 react quantitatively with MgPh2 to afford the C2-symmetric, N-bound Mg amidophosphine complexes rac-7 and (S,S)-7. The former crystallizes as a racemic conglomerate, which is a rare occurrence. Mixing (S,S)- or rac-6 with [IrCl(COE)2]2 leads in both cases to the homochiral dinuclear chloro-bridged P-ligated aminophosphine iridium complexes (S,S,S,S)-9 and rac-9 in excellent yields. X-ray quality single crystals only grow as the racemic compound (or ‘true racemate’) rac-9 thanks to its lowered solubility. In the coordinating solvent CH3CN, rac-9 transforms in high yield into mononuclear Ir-complex rac-10. The crystal structures of compounds rac-6, (S,S)-7, rac-9, and rac-10 reveal the ambidentate nature of the P–N function: amide-coordination in the Mg-complex (S,S)-7 and P-chelation of the softer Ir(I) centres in complexes rac-9 and rac-10. Furthermore, the crystal structures show flexible, symmetry lowering seven-membered P-chelate rings in the Ir complexes and a surprising amount of deformation within the ANDEN backbone. The simulation of this deformation by DFT and SCF calculations indicates low energy barriers. (S,S)-7 and (S,S,S,S)-9 catalyze the intra- and intermolecular hydroamination of alkenes, respectively: 5 mol% of (S,S)-7 affords 2-methyl-4,4′-diphenylcyclopentyl amine quantitatively (7% ee), and 2.5 mol% of (S,S,S,S)-9 in the presence of 5.0 mol% co-catalyst (LDA, PhLi, or MgPh2) gives exo-(2-arylamino)bornanes in up to 68% yield and up to 16% ee.

 Please wait while we load your content...
Please wait while we load your content...