Reversible control of the chromium valence in chemically reduced Cr-doped SrTiO3 bulk powders†
Abstract
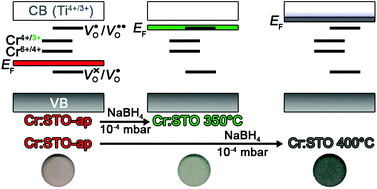
The effect of chemical reduction by NaBH4 on the electronic structure of Cr-doped SrTiO3−δ bulk powders prepared by a solid-state reaction was systematically studied as a function of reduction temperature. Electron paramagnetic resonance (EPR) and diffuse reflectance spectroscopies (DRS) were utilized to monitor changes in the electronic structures of both intrinsic defects (oxygen vacancies and/or Ti3+) and extrinsic dopants (Cr3+) at different reduction temperatures. We identify the existence of two temperature regimes where changes occur within 30 min. The first temperature regime occurs between 300–375 °C and results in (1) reduction of oxygen-related surface defects, and (2) an increase in the concentration of Cr3+ by over an order of magnitude, suggesting that EPR-silent Cr4+ or Cr6+ is being reduced to Cr3+ by NaBH4. The second temperature regime occurs between 375–430 °C where we observe clear evidence of Ti3+ formation by EPR spectroscopy that indicates chemical reduction of the SrTiO3 lattice. In addition, the oxygen-related surface defects observed in regime 1 are not formed in regime 2, but instead lattice oxygen vacancies (VO) are observed by EPR. The changes to the Cr-doped SrTiO3 electronic structure after chemical reduction in regime 1 are quantitatively reversible after aerobic annealing at 400 °C for 30 min. The internal oxygen vacancies formed during the higher temperature reductions in regime 2 require increased temperatures of at least 600 °C to be fully reoxidized in 30 min. The effect of these different oxygen-related defects on the EPR spectrum of substitutional Cr3+ dopants is discussed. These results allow us to independently tune the dopant and host electronic structures of a technologically-relevant multifunctional material by a simple ex situ chemical perturbation.
- This article is part of the themed collection: New Talent: Americas

 Please wait while we load your content...
Please wait while we load your content...