Near-IR luminescent lanthanide complexes with 1,8-diaminoanthraquinone-based chromophoric ligands†
Abstract
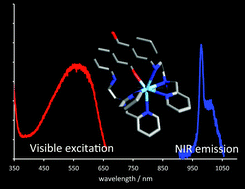
Three new chromophoric anthraquinone-based multidentate ligands have been synthesised in a step-wise manner from 1,8-dichloroanthraquinone. The ligands each comprise two dipicolyl amine units and react with trivalent lanthanide ions to form monometallic complexes of the form [Ln(L)](OTf)3 as indicated by MS studies and elemental analyses. Supporting DFT studies show that the monometallic species are highly favoured (>1000 kJ mol−1) over the formation of a 2 : 2 dimetallic congener. Both ligands and complexes absorb light efficiently (ε ∼ 104 M−1 cm−1) in the visible part of the spectrum, with λabsca. 535–550 nm through an intramolecular charge transfer (ICT) transition localised on the substituted anthraquinone unit. In all cases the complexes show a fluorescence band at ca. 675 nm due to the ICT emitting state. The corresponding Nd(III), Yb(III) and Er(III) complexes also reveal sensitised near-IR emission characteristic of each ion following excitation of the ICT visible absorption band at 535 nm.

 Please wait while we load your content...
Please wait while we load your content...