Role of Cu, Ni and Co metals in the acidic and redox properties of Mo catalysts supported on Al2O3 spheres for glycerol conversion
Abstract
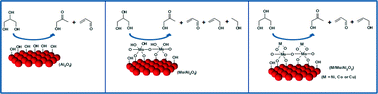
The combined acid–base and redox properties of active Cu, Ni and Co-promoted Mo catalysts supported on Al2O3 spheres have been investigated for the gas-phase dehydration of glycerol. Al2O3 spheres used as supports were impregnated with an aqueous resin containing metal precursors that was synthesized by the polymeric precursor method. The supports and catalysts were characterized by means of chemical analysis, XRD, H2-TPR, N2-physisorption isotherms, SEM-EDS, CO2-TPD, TG/DTA and pyridine adsorption isotherms. The results show differences in morphological, textural and acid–base properties, whereas the XRD and H2-TPR analyses point to a good dispersion of the metal precursors. The H2-TPR analysis shows that the addition of Co, Cu or Ni affects the reducibility of the Mo6+ species, which results in a shift of the H2 consumption peaks to a lower temperature. The addition of Mo resulted in a higher catalytic conversion, particularly for the sample containing Cu (CuMoAl). This improvement in the catalytic performance is possibly related to the presence of active Cu1+/Cu0 and Mo5+/Mo4+ redox species. Although all catalysts favour glycerol dehydration with higher acrolein selectivity, it was found that the redox properties of the Mo species play an important role in the product distribution, which leads to allyl alcohol production with decreased hydroxyacetone yield.

 Please wait while we load your content...
Please wait while we load your content...