Promoting effect of zirconium oxide on Cu–Al2O3 catalyst for the hydrogenolysis of glycerol to 1,2-propanediol†
Abstract
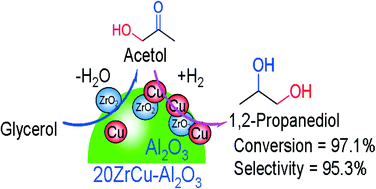
In this work, ZrO2-promoted Cu–Al2O3 catalysts prepared by the co-precipitation method were used for the hydrogenolysis of glycerol to 1,2-propanediol in a fixed-bed reactor. These catalysts were fully characterized by BET, ICP, N2O chemisorption, XRD, H2-TPR, NH3-TPD, XPS, TEM and TGA. The relationship between the catalytic activity and the metal–support interaction was studied in detail. The experimental results showed that the addition of ZrO2 to Cu–Al2O3 could greatly enhance glycerol conversion and 1,2-propanediol selectivity. This improvement was related to the increase in the acidity and Cu dispersion on the catalytic surface. The optimal 20ZrCu–Al2O3 catalyst attained 97.1% glycerol conversion and 95.3% 1,2-propanediol selectivity. Furthermore, the effects of process parameters like solvent, reaction temperature, operating pressure, glycerol concentration and liquid flow rate on glycerol hydrogenolysis together with the catalyst stability were deeply investigated. Compared with the Cu–Al2O3 catalyst, the ZrO2-promoted Cu–Al2O3 catalyst had better stability and potential for practical application, which was likely due to the high Cu dispersion and strong interaction between copper and zirconium species.

 Please wait while we load your content...
Please wait while we load your content...