Chlorophosphines as auxiliary ligands in ruthenium-catalyzed nitrile hydration reactions: application to the preparation of β-ketoamides†
Abstract
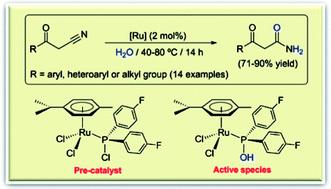
The catalytic hydration of nitriles into amides, in water under neutral conditions, has been studied using a series of arene–ruthenium(II) complexes containing commercially available chlorophosphines as auxiliary ligands, i.e. compounds [RuCl2(η6-p-cymene)(PR2Cl)] (R = aryl, heteroaryl or alkyl group). In the reaction medium, the coordinated chlorophosphines readily undergo hydrolysis to generate the corresponding phosphinous acids PR2OH, which are well-known “cooperative” ligands for this catalytic transformation. Among the complexes employed, best results were obtained with [RuCl2(η6-p-cymene){P(4-C6H4F)2Cl}]. Performing the catalytic reactions at 40 °C with 2 mol% of this complex, a large variety of organonitriles could be selectively converted into the corresponding primary amides in high yields and relatively short times. The application of [RuCl2(η6-p-cymene){P(4-C6H4F)2Cl}] in the preparation of synthetically useful β-ketoamides is also presented.


 Please wait while we load your content...
Please wait while we load your content...