Visible-light-induced photocatalysis and peroxymonosulfate activation over ZnFe2O4 fine nanoparticles for degradation of Orange II†
Abstract
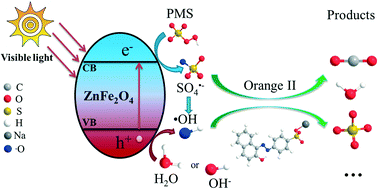
Refractory and non-biodegradable pollutants produced by industries have inevitably brought great threat to human life. Integrating several kinds of advanced oxidation processes (AOPs) into one system has been proposed to be an efficient strategy to remove such pollutants from the environment at low cost. In this study, magnetic zinc ferrite fine nanoparticles, firstly synthesized by a novel soft chemical solution process, showed super reactivity, good reusability and easy separation ability for visible-light-induced Orange II degradation in an integrated ZnFe2O4/PMS (peroxymonosulfate, 2KHSO5·KHSO4·K2SO4, OXONE) aqueous system. Powder X-ray diffraction, transmission electron microscopy, and 57Fe Mössbauer and X-ray photoelectron spectroscopy were employed to characterize the structure and morphology as well as recognize the physicochemical changes of the fine nanoparticles before and after the reaction. The generated oxidizing intermediates during the degradation process were detected by electron paramagnetic resonance spectroscopy and classic quenching experiments, which confirmed that both sulfate radical (SO4˙−) and hydroxyl radical (˙OH) co-existed in the degradation process. The systematic condition experiments further verified the dual functionality of the ZnFe2O4/PMS system, which actively acted as a photocatalyst and a PMS activator for dye molecule oxidation under visible light irradiation. This study proves that photocatalysis and PMS activation for remediation of organic pollutants in water can be easily integrated into one system by using zinc ferrite nanoparticles as an environmentally friendly catalyst.

 Please wait while we load your content...
Please wait while we load your content...