Tuning the selectivity toward CO evolution in the photocatalytic conversion of CO2 with H2O through the modification of Ag-loaded Ga2O3 with a ZnGa2O4 layer†
Abstract
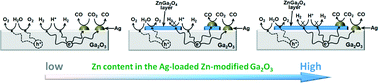
Stoichiometric evolutions of CO, H2, and O2 were achieved for the photocatalytic conversion of CO2 with H2O as an electron donor using Ag-loaded Zn-modified Ga2O3. The selectivity toward the evolution of CO over H2 can be controlled by varying the amount of Zn species added in the Ag-loaded Zn-modified Ga2O3 photocatalyst. The production of H2 gradually decreased with increasing amounts of Zn species from 0.1 to 10.0 mol%, whereas the evolution of CO was almost unchanged. The XRD, XAFS, and XPS measurements revealed that a ZnGa2O4 layer was generated on the surface of Ga2O3 by modification with Zn species. The formation of the ZnGa2O4 layer eliminated the proton reduction sites on Ga2O3, although the crystallinity, surface area, and morphology of Ga2O3 as well as the particle size and chemical state of Ag did not change. In conclusion, we designed a highly selective photocatalyst for the conversion of CO2 with H2O as an electron donor using Ag (the cocatalyst for the CO evolution), ZnGa2O4 (the inhibitor of the H2 production), and Ga2O3 (the photocatalyst).

 Please wait while we load your content...
Please wait while we load your content...