Steric confinement and enhanced local flexibility assist knotting in simple models of protein folding†
Abstract
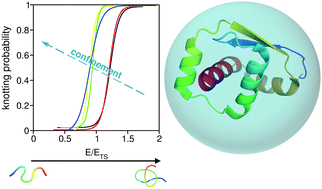
The chaperonin complex GroEL–GroES is able to accelerate the folding process of knotted proteins considerably. However, the folding mechanism inside the chaperonin cage is elusive. Here we use a combination of lattice and off-lattice Monte Carlo simulations of simple Gō models to study the effect of physical confinement and local flexibility on the folding process of protein model systems embedding a trefoil knot in their native structure. This study predicts that steric confinement plays a specific role in the folding of knotted proteins by increasing the knotting probability for very high degrees of confinement. This effect is observed for protein MJ0366 even above the melting temperature for confinement sizes compatible with the size of the GroEL/GroES chaperonin cage. An enhanced local flexibility produces the same qualitative effects on the folding process. In particular, we observe that knotting probability increases up to 40% in the transition state of protein MJ0366 when flexibility is enhanced. This is underlined by a structural change in the transition state, which becomes devoid of helical content. No relation between the knotting mechanism and flexibility was found in the context of the off-lattice model adopted in this work.


 Please wait while we load your content...
Please wait while we load your content...