Electron transport via a soluble photochromic photoreceptor†
Abstract
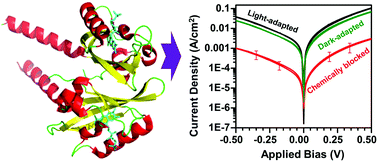
Electron transport properties via a photochromic biological photoreceptor have been studied in junctions of monolayer assemblies in solid-state configurations. The photoreceptor studied was a member of the LOV domain protein family with a bound flavin chromophore, and its photochemically inactive mutant due to change of a crucial cysteine residue by a serine. The photochemical properties of the protein were maintained in dry, solid state conditions, indicating that the proteins in the junctions were assembled in native state-like conditions. Significant current magnitudes (>20 μA at 1.0 V applied bias) were observed with a mechanically deposited gold pad (area ∼0.002 cm2) as top electrode. The current magnitudes are ascribed to electrode–cofactor coupling originating from the apparent perpendicular orientation of the protein's cofactor embedded between the electrodes, and its proximity to the electrodes. Temperature independent electron transport across the protein monolayers demonstrated that solid-state electron transport is dominated by tunneling. Modulation of the observed current by illumination of the wildtype protein suggested conformation-dependent electron conduction efficiency across the solid-state protein junctions.


 Please wait while we load your content...
Please wait while we load your content...