Organic–inorganic interactions of single crystalline organolead halide perovskites studied by Raman spectroscopy†
Abstract
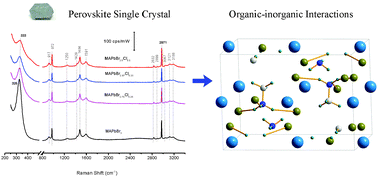
Organolead halide perovskites exhibit superior photoelectric properties, which have given rise to the perovskite-based solar cells whose power conversion efficiency has rapidly reached above 20% in the past few years. However, perovskite-based solar cells have also encountered problems such as current–voltage hysteresis and degradation under practical working conditions. Yet investigations into the intrinsic chemical nature of the perovskite material and its role on the performance of the solar cells are relatively rare. In this work, Raman spectroscopy is employed together with CASTEP calculations to investigate the organic–inorganic interactions in CH3NH3PbI3 and CH3NH3PbBr3−xClx perovskite single crystals with comparison to those having ammonic acid as the cations. For Raman measurements of CH3NH3PbI3, a low energy line of 1030 nm is used to avoid excitation of strong photoluminescence of CH3NH3PbI3. Raman spectra covering a wide range of wavenumbers are obtained, and the restricted rotation modes of CH3–NH3+ embedded in CH3NH3PbBr3 (325 cm−1) are overwhelmingly stronger over the other vibrational bands of the cations. However, the band intensity diminishes dramatically in CH3NH3PbBr3−xClx and most of the bands shift towards high frequency, indicating the interaction with the halides. The details of such an interaction are further revealed by inspecting the band shift of the restricted rotation mode as well as the C–N, NH3+ and CH3 stretching of the CH3NH3+ as a function of Cl composition and length of the cationic ammonic acids. The results show that the CH3NH3+ interacts with the PbX3− octahedral framework via the NH3+ end through N+–H⋯X hydrogen bonding whose strength can be tuned by the composition of halides but is insensitive to the size of the organic cations. Moreover, an increase of the Cl content strengthens the hydrogen bonding and thus blueshifts the C–N stretching bands. This is due to the fact that Cl is more electronegative than Br and an increase of the Cl content decreases the lattice constant of the perovskite. The findings of the present work are valuable in understanding the role of cations and halides in the performance of MAPbX3-based perovskite solar cells.

 Please wait while we load your content...
Please wait while we load your content...